MindMap Gallery The Effect of Temperature on Reaction Rate
- 25
The Effect of Temperature on Reaction Rate
The effect of temperature on reaction rate involves increasing rates with higher temperatures due to more energetic reactants, collision theory explaining reactions occur via collisions with sufficient energy and orientation, and activation energy required for reactants to reach a transition state, with lower energy leading to faster rates.
Edited at 2024-12-23 07:27:47- Compound: How to use elastin
In the introduction section, elastin is a protein that plays a crucial role in the body, endowing tissues with elasticity and toughness. Its properties include high elasticity and good ductility. Elastin has a wide range of sources and is commonly found in animal connective tissues such as skin and blood vessels. When using elastin in products, the amount and method of addition should be determined based on the characteristics of the product. It has many uses and can be added to skincare products to enhance skin elasticity and reduce wrinkles; Improving food texture and enhancing taste in the food industry; In the medical field, it can be used to make elastic scaffolds such as artificial blood vessels, providing support for tissue repair. Reasonable use can play its unique value.
- Compound: Instructions for using protein
This document aims to guide the proper use of proteins. Proteins are the fundamental substances that make up life and are essential for maintaining normal physiological functions in the body, with a wide variety of types. When using protein, daily diet is an important source, such as meat, eggs, etc., which are rich in high-quality protein. Measuring protein intake can be done using professional tools or methods to ensure that the intake meets the body's needs. When mixing protein powder, it should be prepared according to the recommended ratio, and the water temperature should not be too high to avoid damaging the protein activity. Different groups of people have different protein requirements, such as athletes and fitness enthusiasts who can increase their intake appropriately to meet the needs of body repair and muscle growth.
- Compound: Usage of Unsaturated Polyester Resin
Introduction to Unsaturated Polyester Resin: It is an important thermosetting resin with various excellent properties. It has a wide range of applications and is used in the construction industry to produce fiberglass products such as doors, windows, decorative panels, etc; Used in the automotive industry for manufacturing body components. The advantages of use include simple molding process, low cost, and chemical corrosion resistance. However, it also faces challenges and limitations, such as relatively poor heat resistance and susceptibility to aging. During use, corresponding protective measures should be taken according to specific application scenarios, such as adding heat-resistant agents, antioxidants, etc., to extend their service life and fully leverage their advantages.
The Effect of Temperature on Reaction Rate
- Compound: How to use elastin
In the introduction section, elastin is a protein that plays a crucial role in the body, endowing tissues with elasticity and toughness. Its properties include high elasticity and good ductility. Elastin has a wide range of sources and is commonly found in animal connective tissues such as skin and blood vessels. When using elastin in products, the amount and method of addition should be determined based on the characteristics of the product. It has many uses and can be added to skincare products to enhance skin elasticity and reduce wrinkles; Improving food texture and enhancing taste in the food industry; In the medical field, it can be used to make elastic scaffolds such as artificial blood vessels, providing support for tissue repair. Reasonable use can play its unique value.
- Compound: Instructions for using protein
This document aims to guide the proper use of proteins. Proteins are the fundamental substances that make up life and are essential for maintaining normal physiological functions in the body, with a wide variety of types. When using protein, daily diet is an important source, such as meat, eggs, etc., which are rich in high-quality protein. Measuring protein intake can be done using professional tools or methods to ensure that the intake meets the body's needs. When mixing protein powder, it should be prepared according to the recommended ratio, and the water temperature should not be too high to avoid damaging the protein activity. Different groups of people have different protein requirements, such as athletes and fitness enthusiasts who can increase their intake appropriately to meet the needs of body repair and muscle growth.
- Compound: Usage of Unsaturated Polyester Resin
Introduction to Unsaturated Polyester Resin: It is an important thermosetting resin with various excellent properties. It has a wide range of applications and is used in the construction industry to produce fiberglass products such as doors, windows, decorative panels, etc; Used in the automotive industry for manufacturing body components. The advantages of use include simple molding process, low cost, and chemical corrosion resistance. However, it also faces challenges and limitations, such as relatively poor heat resistance and susceptibility to aging. During use, corresponding protective measures should be taken according to specific application scenarios, such as adding heat-resistant agents, antioxidants, etc., to extend their service life and fully leverage their advantages.
- Recommended to you
- Outline
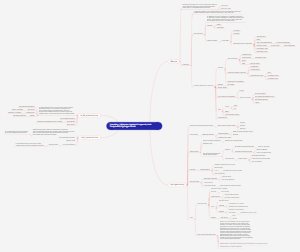
The Effect of Temperature on Reaction Rate
Definition of Reaction Rate
Measure of change in concentration of reactants/products over time
Temperature's Influence
Increase in Temperature
Higher Kinetic Energy
Molecules move faster
More frequent collisions
Greater Collision Energy
Higher likelihood of effective collisions
Effective collisions lead to reaction
Decrease in Temperature
Lower Kinetic Energy
Molecules move slower
Less frequent collisions
Lower Collision Energy
Fewer effective collisions
Slower reaction rate
Collision Theory
Collision Frequency
Directly proportional to temperature
Energy of Colliding Particles
Must exceed activation energy for reaction
Activation Energy
Minimum energy required for a reaction to occur
Higher temperature increases fraction of molecules with sufficient energy
Experimental Observations
Reaction Rate vs. Temperature Plots
Arrhenius Equation
Describes the relationship between temperature and reaction rate
ln(k) vs. 1/T plot yields a straight line
Slope related to activation energy
Practical Implications
Industrial Processes
Optimizing temperature for faster reactions
Energy Consumption
Balancing reaction rate with energy costs
Chemical Stability
Temperature control to prevent unwanted reactions
Factors Affecting Temperature's Impact
Nature of Reactants
Different substances have different activation energies
Presence of Catalysts
Lower activation energy, increase reaction rate at lower temperatures
Concentration of Reactants
Higher concentrations can increase collision frequency
Pressure (for gases)
Can increase collision frequency by reducing volume
Surface Area (for solids)
Increased surface area leads to more collisions with reactants
Mathematical Representation
Arrhenius Equation
k = Ae^(Ea/RT)
k is the rate constant
A is the preexponential factor
Ea is the activation energy
R is the gas constant
T is the temperature in Kelvin
Van't Hoff's Rule
Doubling the temperature increases reaction rate by a factor of 24 for every 10°C rise
Approximate rule of thumb for many reactions